Introduction
PhyTip® columns from Biotage® provide the ideal technology for high-throughput protein purification on the CyBio FeliX liquid handler from Analytik Jena. Each PhyTip® contains an affinity resin bed which captures the protein, keeps it bound through successive wash cycles and releases purified protein during elution. A range of affinity resins are available which selectively immobilise specifically tagged proteins and antibodies. Hence, the combination of PhyTips® with the CyBio FeliX provides a universal platform for the purification of a wide range of proteins from bacterial and eukaryotic cell lysates.
This application note describes the purification of histidinetagged proteins using PhyTip® columns, filled with an immobilised metal affinity chromatography (IMAC) resin.
PhyTips® were used with the CyBio FeliX, a small footprint 12-position liquid handler with a volume range between 1-1000 µL. The PhyTip® columns were loaded with the bacterial lysates containing recombinantly expressed histidine-tagged proteins and extraction buffer. After 120 minutes, protein samples had been washed and eluted into a collection plate. Protein concentration was assessed by Bradford assay, and purity via sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The identity of purified protein was then confirmed via mass spectrometry.
The CyBio FeliX in combination with PhyTips® provides a fast and convenient method to purify proteins at high concentration and purity. This fully walk away method can be tailored to target a range of protein features. The rapid method can then be scaled to process as many purifications as needed.
Materials and Methods
Labware and reagentsBacterial Growth and Lysis- BL21(DE3) Singles Competent Cells – Novagen, SigmaAldrich #70235-M
- Ampicillin – Merck® #A8351-25G
- Auto induction media – Formedium #AIMTB0210
- BugBuster® Master Mix – Merck® #71456
- Nunc 96 Well Polypropylene DeepWell Storage Plates – Thermo Fisher #278743
Protein purification- 1000 µL PhyTip® columns, with 10 µL Ni-IMAC affinity resin - Biotage® #PTT-91-10-03
- 1000 µL PhyTip® columns, with 160 µL Ni-IMAC affinity resin - Biotage® #PTT-91-16-03
- 5x IMAC equilibration & capture buffer, 25 mM imidazole
- 5x IMAC wash Buffer: phosphate buffer, 25 mM imidazole
- 1x IMAC elution Buffer: 10 mM NaH2PO4, 0.3 M NaCl and 200 mM imidazole, pH7.4
- 5x Nunc 96-Well Polypropylene DeepWell Storage Plates – Thermo Fisher #278743
- 3x Axygen Single Well High Profile Reagent Reservoirs – Corning #RES-SW96-HP-SI
- 2x CyBio TipRack 96/1000 µL – Analytik Jena#OL3811-25-539-N
- 3x CyBio Support 97 mm – Analytik Jena #OL3317-11-105
- 3x CyBio RoboTipTray 1-96/1000 µL #OL3810-24-023
Protein quantification- Bovine serum albumin (BSA) – New England Biolabs #B9000S
- Bradford Reagent – Merck® #B6916-500ML
- Greiner flat bottom 96-Well Plate, polystyrene – Greiner #65516
SDS gel electrophoresis- Bolt 4-14% Bis-Tris Plus Gel – Invitrogen #NW04120BOX
- Page Ruler Plus Prestained Protein Ladder – Thermo Fisher #26619
- InstantBlue® Coomassie Protein Stain – Abcam #ab119211
- 4X Bolt LDS Sample Buffer – Thermo Fisher #B0008
- 10X Bolt Sample Reducing Agent – Thermo Fisher B0009

DNA purification- E.Z.N.A.® Plasmid DNA Mini Kit II –Omega BIO-TEK #D6945-002
- Isopropanol - Merck® #109634
- Ethanol - VWR® #20821.365
Protein digestion and identification- Ammonium bicarbonate – Merck® #101131
- Dithiothreitol – Merck® #111474
- Iodoacetoamide – Merck® #804744
- Trypsin, porcine, sequencing grade, modified – Promega Corp #9PIV511
- Water, HPLC grade – Merck® #V270733
- Acetonitrile, HPLC gradient grade – Merck® #100030
- Formic acid, HPLC – Merck® #543804
- Trifluoroacetic acid – Sigma #302031
Instruments
- CyBio FeliX Basic Unit with Enclosure – Analytik Jena #OL5015-24-100
- CyBio FeliX Head R 96/1000 µL – Analytik Jena #OL3316-14-950
- CLARIOstar® Plus microplate reader – BMG Labtech
- iBright FL1500 Imaging System– Thermo Fisher #A44241
MethodsProteins, recombinantly expressed in BL21 (DE3) Escherichia coli strains were selected for purification with PhyTip® columns. The expression plasmid peT21a contained a T7-Lacl promoter, and a C-terminal 6 poly histidine-tag. The histidine-tagged protein has an estimated size of 27.6 kDa.
Growth and lysisExpression cultures were inoculated from glycerol stocks in auto induction media, supplemented with 100 µg/mL of ampicillin. After incubation at 37 °C for 16 hours at 200 rpm shaking, bacteria were harvested by centrifugation at 2,500 xg for 20 minutes at 4 °C. The supernatant was discarded, and 1 mL of lysis solution was added to the
bacterial pellets. Pellets were resuspended in lysis buffer by placing them on an orbital shaker for 30 minutes at 300 rpm. Lysed cells were centrifuged again at 21,000 xg for 20 minutes. One milliliter of cleared lysate was transferred to a 96 deep well plate (#278743) ready for automated protein purification.
Plasmid isolation and sequencingThe plasmid sequence was confirmed prior to PhyTip® purification. Plasmid DNA was isolated from bacterial cultures using the Omega BIO-TEK E.Z.N.A.® Plasmid DNA
Mini Kit II, as per manufacturer’s instructions. Ten microliters of plasmid DNA was then submitted to Source BioScience for Sanger Sequencing. Plasmid was sequenced with the following primers at 3.2 pmol/µL; Forward TAA TAC GAC TCA CTA TAG GG, Reverse GCT AGT TAT TGC TCA GCG G.
Protein purification with the CyBio FeliXEquilibration and wash buffers provided with the PhyTip® column kit at 5x concentration were diluted to 1x concentration using dH2O. Elution buffer was provided at 1x concentration. All three buffers were decanted into single well reservoirs (#RES-SW96-HP-SI). The required volume of each buffer depends upon the number of PhyTip® columns in the automated run.
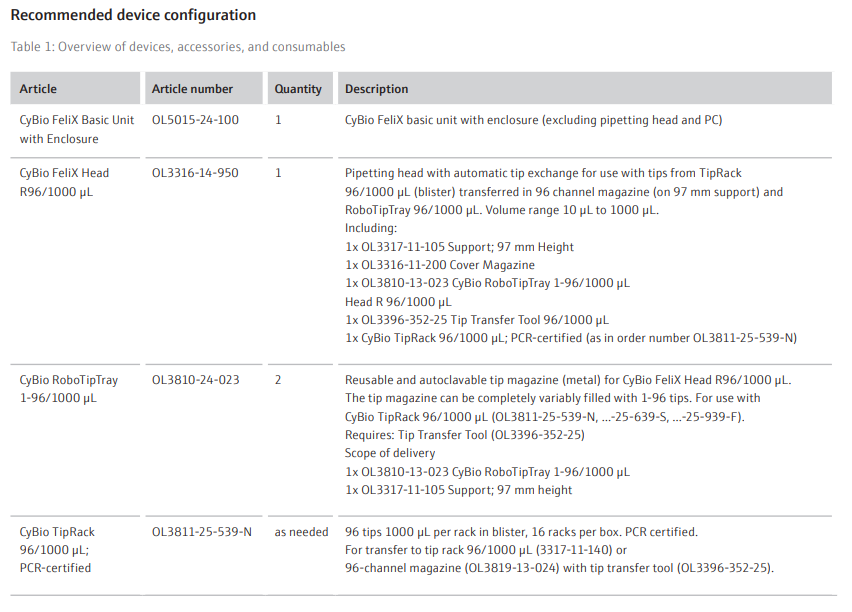
The labware and pipetting tips required for the protein purification were placed onto the CyBio FeliX, as shown in Figure 2. Once the CyBio FeliX deck was loaded, the
purification program was initiated, and the robot performed the pipetting steps shown below.
1. Buffer Transfer: Buffers provided in single well reservoirs were aliquoted to 96 deep well plates using 1000 µL tips.
2. Equilibration: PhyTip® columns were equilibrated through multiple pipetting cycles of the equilibration buffer, priming the resin.
3. Protein Capture: PhyTip® columns aspirated and dispensed the bacterial lysate multiple times, to bind the target proteins from the lysate to the resin.
4. Wash: PhyTip® column resin was washed through aspirating and dispensing of wash buffer, through two separate plates.
5. Elution: Proteins bound to PhyTip® columns were eluted via pipetting cycles in elution buffer of 100 µL. This elution step was repeated in a second elution plate with 100 µL.
Once the program was completed, purified proteins eluted into two 96 well plates were removed from the instrument for downstream processing. The tips used to aliquot wash and equilibration buffer (Step 1) can be re-used in successive purifications.
 Protein quantification and purity assessment
Protein quantification and purity assessmentProtein concentration was determined using a Bradford standard curve of bovine serum albumin (BSA). Six concentrations of BSA protein standards were prepared at the
following concentrations: 0.1, 0.2, 0.4, 0.6, 1.0, and 1.4 mg/mL. In a flat bottom 96 well plate, 5 µL of each protein standard and 5 µL of PhyTip® purified proteins were added to separate wells. A blank well with 5 µL elution buffer provided with the PhyTip® kit was also added to the plate. Finally, 250 µL of 1x Bradford reagent was added to each well and after incubation for 20 minutes in the dark at room temperature, absorbance at 595 nm was measured.
Proteins from the PhyTip® purification were also analysed via SDS-PAGE, along side the starting bacterial lysate. To 1.6 µL of sample, 10 µL of sample buffer, 4 µL of reducing agent, and 24.4 µL dH2O was added. Samples were denatured at 70 °C for 10 minutes. A total of 40 µL of samples were analysed by SDS-PAGE including 5 µL of ladder. Samples were run for 22 minutes at 200 V. Finally, gels were stained for 60 minutes in instant blue stain before destaining for 30 minutes in dH2O. Protein bands were visualized with the iBright imaging system.
Protein Identification and purity assessmentIntact proteins in solution were first characterised by LC/MS using an Agilent PLRP-S column and a gradient of 20-50% acetonitrile, with ESI-Q-ToF mass analysis. Spectra were acquired over the m/z range 300-3,200, with protein mass determined by deconvolution of the raw mass spectra.
In addition, protein bands separated via SDS-PAGE were isolated and then submitted for proteomics analysis as described in Shevchenko et al.[1], to confirm they corresponded to the target protein. In brief, the most intense protein bands corresponding to molecular weights of 10, 25, and 30 kDa from purified samples were excised and centrifuged. These gel slices were incubated with ammonium bicarbonate buffer and acetonitrile to remove the protein stain. Gel slices were then saturated with trypsin to generate peptides. These peptides were identified by LC/MS/MS using an ESI-Q-ToF operating in auto-MS/MS mode. Peptides were eluted from an Agilent AdvanceBio Peptide column with a 2-40% acetonitrile gradient over 12.5 minutes. The accumulated spectra were searched against either a SwissProt database or custom sequences using SpectrumMill (Agilent).
Results and Discussion
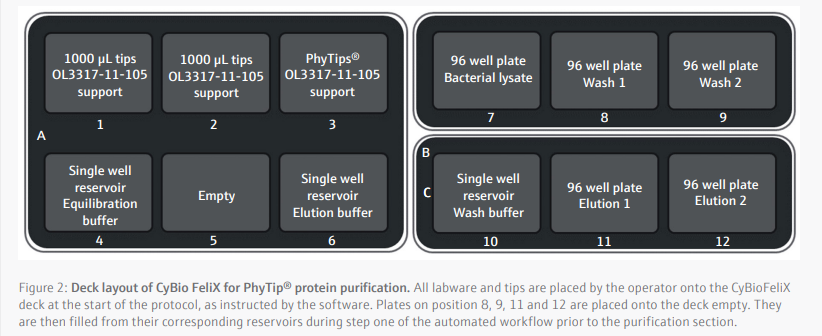
Protein yieldThe histidine-tagged proteins with an estimated size of 27.6 kDa were purified with PhyTip® columns containing either 10 or 160 µL of purification resin. Protein concentration was assessed via Bradford assay (Figure 3).
Protein yield was correlated to the total volume of PhyTip® resin. For the 10 µL resin tips, an average of 1.0 mg/mL was purified in 200 µL of elution buffer. Most of the protein, 1.7 mg/mL , was eluted in the first elution stage, with the second elution containing only 0.3 mg/mL protein. For the 160 µL tips, an average of 2.0 mg/mL was purified in 200 µL of elution buffer. With the larger resin tips, the concentration of the second elution was 1.7 mg/mL, 76% of the first elution. Additional protein may be recovered through additional elution steps or larger elution volumes.
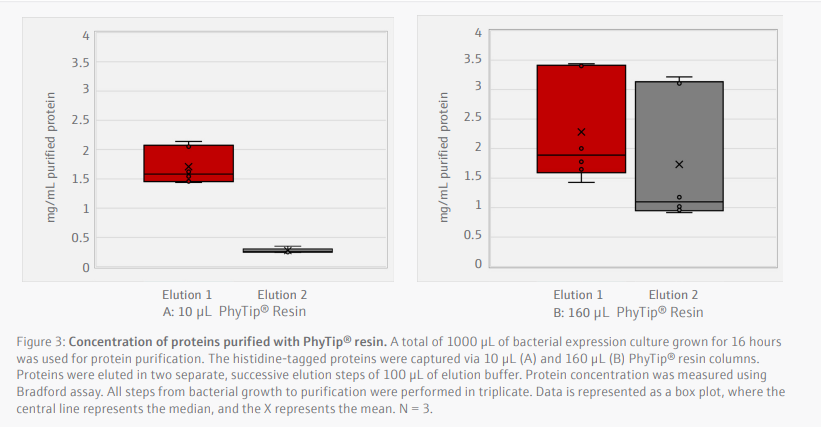
Purified proteins eluted in the first and second stages with the 10 µL resin tips were loaded onto an SDS-PAGE (Figure 4). Three major bands were identified around 10, 25, and 30 kDa. Mass spectrometry analysis identified the bands at 25 and 30 kDa corresponded to the protein sequence of the target. The lower band at 10 kDa corresponded to an E. coli outermembrane lipoprotein, also present in the bacterial lysate.
 Discussion
DiscussionUsing PhyTip® columns on the CyBio FeliX enables the rapid purification of proteins in a high-throughput format. The yield and final concentration of protein recovered correlates with the quantity of PhyTip® resin used for the purification. Additional purified protein can be recovered by increasing the elution volume as desired.
The purity of protein purification was assessed with SDS-PAGE. The most abundant protein band isolated with PhyTip® columns corresponded to the expected weight of 28 kDa. A likely truncated isoform was also identified, which is similarly present in the bacterial lysate. Addition of protease inhibitors in the lysis buffer could reduce proteolysis, minimizing secondary bands. Other bands present are likely endogenous E. coli proteins with native histidine residues clusters, commonly seen in purification approaches targeting histidine-tagged proteins (Robichon et al., 2011)[2].
Summary
High-throughput protein purification remains a technically challenging and time-consuming process. With PhyTip® columns, protein targets can be purified with high yields and purity in a few simple steps, with limited assay optimisation required. In combination with the CyBio FeliX, automated PhyTip® columns permit the processing of 96 protein purifications in just under 2 hours. The fully automated workflow improves reproducibility between sample extractions, while freeing up researcher time for downstream applications.
Automation of PhyTip® columns with the CyBio FeliX is the ideal solution for high-throughput protein purification in a compact and powerful format. Pre-written methods are available for the CyBio FeliX to perform PhyTip® purifications in an accessible and flexible format.